
 1, Nadia Pieretti
1, Nadia Pieretti
 2, Robert John Young
2, Robert John Young
 3, Marina Duarte
3, Marina Duarte
 1*
1*https://doi.org/10.21068/c2021.v22n01a06
Sara Gonçalves Santos
 1, Nadia Pieretti
1, Nadia Pieretti
 2, Robert John Young
2, Robert John Young
 3, Marina Duarte
3, Marina Duarte
 1*
1*
Received: April 4, 2020
Accepted: November 9, 2020
Anthropogenic noise, which is part of an urban soundscape, can negatively affect the behaviour of wild animals. Here we investigated how biophony (animal sounds) was affected by noise in an urban Brazilian forest fragment. Our hypothesis was that noise and biophony would differ between the border and the centre of the forest fragment (i.e., lower biophony predicted in noisy areas). Two passive acoustic monitoring devices were used to record soundscapes one week per month, 24 hour per day, from May to July 2012. The Acoustic Complexity Index (ACI) was used to quantify biophony and the Power Spectral Density (PSD) to quantify urban noise. PSD and ACI were higher on the border than in the centre of the fragment. PSD was lower in July, while the ACI did not significantly vary between months. Noise levels were also higher on the border. Conversely, potential species richness was higher in the centre of the forest fragment. Higher biophony at noisy sites can be interpreted as behavioural responses of species for communicating in noisy areas. Alternatively, they could be the result of species segregation by degree of vocal plasticity or due to differences in composition of communities.
Keywords. Animal communication. Ecoacoustics. Noise. Soundscapes.Urban environment.
El ruido antropogénico, que forma parte de un paisaje sonoro urbano, puede afectar negativamente el comportamiento de los animales. En este estudio investigamos cómo la biofonía (sonidos de animales) se vió afectada por el ruido antropogénico en un fragmento de bosque urbano brasileño. Nuestra hipótesis es que el ruido y la biofonía difiren entre el borde y el centro del fragmento de bosque (es decir, una menor biofonía en áreas ruidosas). Se usaron dos dispositivos de monitoreo acústico pasivo para grabar paisajes sonoros una semana al mes, 24 horas al día, de mayo a julio de 2012. El índice de complejidad acústica (ACI) se usó para cuantificar la biofonía y la densidad espectral de potencia (PSD) para cuantificar el ruido urbano. Se obtucieron mayores valores de PSD y ACI en el borde que en el centro del fragmento. La PSD fue menor en julio, mientras que el ACI no varió significativamente entre meses. Los niveles de ruido también fueron más altos en el borde, mientras que la riqueza potencial de especies fue mayor en el centro del fragmento de bosque. Una mayor biofonía en áreas ruidosas puede ser interpretada como el efecto de respuestas conductuales de las especies con el fin de establecer una comunicación efectiva. Alternativamente, podrían ser el resultado de la segregación de especies por grado de plasticidad vocal o debido a diferencias en la composición de las comunidades.
Palabras clave. Ambiente urbano. Comunicación animal. Ecoacústica. Ruido. Paisajes sonoros.
Several studies have shown that noise pollution can affect the behaviour, reproduction and survival of wild animals living in cities (Slabbekoorn & Peet, 2003; Warren et al., 2006; Bonier et al., 2007; Duarte et al., 2011; Santos et al., 2017). However, most of these studies are experimental and made under controlled conditions, usually with a single species (Berger-Tal et al., 2019). Few studies have addressed the impact of noise pollution at a broader ecological level, the community, by using an ecoacoustics approach. Ecoacoustics investigates all the sound sources (the soundscape) as a means conveying important information about the ecological status of ecosystems (Sueur & Farina, 2015).
According to Schafer (1977), sounds are ecological properties of landscapes, and soundscapes are the acoustic characteristics of an area that reflect natural and anthropogenic processes. A soundscape is formed by three acoustic components: biophony (biological sounds such as animal vocalizations), geophony (natural non-biological sounds, such as that produced by wind, rain, and thunder) and anthropophony (sounds generated by humans such as traffic noise; Pijanowski et al., 2011). The study of soundscapes can provide valuable information on animal communication dynamics, help the assessment of the environmental status of habitats and their spatiotemporal variations, and investigate the noise effects on different ecosystems (Pieretti et al., 2011).
All habitats have some level of anthropogenic noise, but urban sounds produced by cars, motorcycles, trains, and airplanes, in addition to the sounds produced in buildings and industries, are significantly different from most natural sounds because most of their energy is concentrated in low frequencies (below 2000 Hz) and has longer duration (Warren et al., 2006; Brumm, 2006; Slabbekoorn & Ripmeester, 2008). High noise levels can mask animal acoustic signals, such as sounds from reproductive partners, alarm calls, parental care and territorial defense songs (Brumm et al., 2004; Santos et al., 2017). The masking effect can force species to use compensatory mechanisms to vocally communicate or abandon noisy areas (Nemeth & Brumm, 2009; Santos et al., 2017). A common behavioural adaptation employed to overcome masking consists in increasing the amplitude of calls, a response known as Lombard Effect (Brumm & Zollinger, 2011). Additionally, many species have been found to change the features of the calls, such as frequency, duration, number of notes in noisy places (Warren et al., 2006; Duarte et al., 2017; Tolentino et al., 2018). Studies on the impact of noise pollution on animals are important because they can drive the elaboration of management strategies and conservation of urban forests, which are wildlife refuges (Barber et al., 2009; Duarte et al., 2011, Teixeira et al., 2015; Santos et al., 2017).
In the present study, we investigated the soundscape comparing biophony and noise levels (i.e., sound pressure levels) between the border and the centre of a tropical urban forest fragment. We also identified and quantified the number of species calling at both sites. Our hypothesis was that noise and biophony levels would differ significantly between the two sites. We predicted higher levels of biophony and potential species richness where noise was lower.
General considerations on passive acoustic monitoring (PAM).
Passive acoustic monitoring is a method used in the study of soundscape ecology, both in terrestrial and aquatic environments. Acoustic sensors can record sounds for prolonged periods and produce a massive amount of data. Several acoustic indexes have been developed to optimize data analysis and extract relevant ecological information from such recordings. Among them, the acoustic complexity index (ACI) has the purpose of quantifying biophony by processing the intensities of the vocalizations recorded in audio files, even in the presence of continuous anthropogenic noise (Pieretti et al., 2011). By providing an estimate of the amount of biophony in an environment, the ACI has proven to be a useful tool to evaluate behavioural changes and define the composition of an animal community (Pieretti et al., 2011; Farina et al., 2011a).
Study area. This study was conducted in an urban tropical forest fragment located inside the campus of the Pontifical Catholic University (PUC) of Minas Gerais, in the Northwest Zone of Belo Horizonte, Minas Gerais, Brazil (19º55'10 S 43º59'31 W; Figure 1). The elevation of the area ranges from 870 to 930 m above sea level. The PUC forest is a remnant fragment of Atlantic forest of 66 755 m², characterized as a semideciduous seasonal secondary forest with many species of insects, anurans, reptiles, birds (approximately 134 species, see Vasconcelos et al., 2013) and mammals. Currently, this area is surrounded by a densely anthropized urban matrix, which hampers or prevents the dispersal of many species to other fragments. A museum of Natural Sciences, a sport centre and a small airport (Aeroporto Carlos Prates in activity since 1944) are located in the area surrounding the forest fragment (i.e. within 2 km radius).
Data collection. The data were collected using two passive acoustic monitoring sensors (Song Meter Digital Field Recorder -SM2- Wildlife Acoustics, Inc., USA) installed at 1.5 m height above ground level in the central area (points A and B) and other two on the border of the forest fragment (points C and D; Figure 1). To prevent overlap of the sound recorded (i.e., to ensure independence of data sources), the distance between each SM2s in the centre and border stations was 80 meters. The distance between the stations (centre and border) was approximately 300 meters. The distances between the SM2s and roads were 30 meters for the border station and 100 meters for the centre station (Figure 1). Similar distances were tested and published in our previous studies in Atlantic forest areas (Duarte et al., 2015). SM2 were configured to record 24 hours per day, one week per month, during three consecutive months, from May to July 2012. In May, data were collected between the 24th and 31st, in June from the 21st to the 28th, and in July from the 24th to the 31st. The devices recorded by using a sampling rate of 44.1 kHz, 36 dB microphone gain, wave format files and on stereo channel using two omnidirectional waterproof microphones with a flat frequency response of 0.020–20 kHz, sensitivity of -36 ± 4 dB and a signal to noise ratio of >62 dB.
We also conducted two 15-minute measurements of the background sound pressure levels once per month at each recording point, using a Z-weighted B&K2270 sound level meter. All the animal sounds close to the microphone were excluded from the recordings using the BZ5503 software (Bruel and Kjaer, Denmark). The equivalent sound levels (Leq), which are the standard for sound-pollution measurements, were then calculated (Rossing, 2007).
Together with noise, species richness and species abundance could influence acoustic differences between border and centre of the forest fragment. Unfortunately, abundance is difficult to evaluate using PAM (Duarte et al., 2015). Therefore, potential species richness was calculated for each site using aural identification (i.e., manual) of animal sounds. This analysis was done using spectrograms created in Raven Pro 1.5 software. One day (24 hours) per recording session was randomly selected from each sampling point for species’ identification surveys. Sounds emitted by insects, anurans, birds and mammals were identified by taxon group specialists who visually and aurally inspected the first three-minutes of every quarter hour. Where the sounds could not be associated to a particular species, they were classified as sound types in order to determine the potential number of species at each site.

Figure 1. Sampling points for biophony at the Pontifical Catholic University forest fragment, Belo Horizonte, Minas Gerais, Brazil. A and B represent the centre, and C and D the forest border.
Data analysis. All files were processed via the ACI (Pieretti et al. 2011) to obtain a measure of biophony. Anthropogenic noise was quantified using the power spectral density (PSD). ACI and PSD were extracted using Wavesurfer software (Sjölander & Beskow, 2000).
The ACI metrics, like other indices created to operate in soundscape analysis, are based on the fact that there is a strict and direct relationship between the complexity of animal communities, and the spectral and temporal complexity of a soundscape. In other words, the acoustic information expressed by the ACI is greater in a location where there are more individuals and/or more species. (Farina et al., 2011b). The ACI algorithm has already been successfully applied in terrestrial habitats (Pieretti & Farina, 2013; Bobryk et al., 2015), including forest fragments in neotropics (Pieretti et al., 2015; Duarte et al., 2015).
The power spectral density is a quantitative measure of the acoustic energy of the environmental sounds expressed per unit frequency (Duarte et al., 2015). It is commonly used to obtain the distribution of the power across the frequency domain. At low frequencies, the acoustic energies at our study sites were mainly driven by urban noise pollution. Consequently, the PSD was used as an indicator of anthropogenic noise.
The sampling points were grouped in central area (points A and B) and border area (points C and D). Recordings were subsampled analyzing three minutes of every fifteen minutes, totalizing 403.2 hours. To calculate the ACI, the Fast Fourier Transform - FFT of 512 points was used (frequency bin: 86Hz; temporal step: 0.01s). A one-second grouping value was used, in order to obtain one ACI value at every second for every frequency bin, successively averaged over the lenght of the recording (three minutes). Sounds concentrated below 1550 Hz were measured by summing PSD values in the relative frequency bins (expressed in V2 Hz−1). ACI values (from 1550 to 16 000 Hz) were considered as biophony, emitted by insects, birds and mammals (Figure 2). Temporal distribution of both PSD and ACI were carried out.
The data of ACI and PSD did not present normal distributions. Therefore, a log transformation was performed to remove the effects of magnitude differences between variables, avoiding negative numbers, normalizing the data and increasing the importance of the smaller values (Manly, 1997). Subsequently, Pearson’s correlations and ANOVA tests (Dunn post hoc test) were performed.
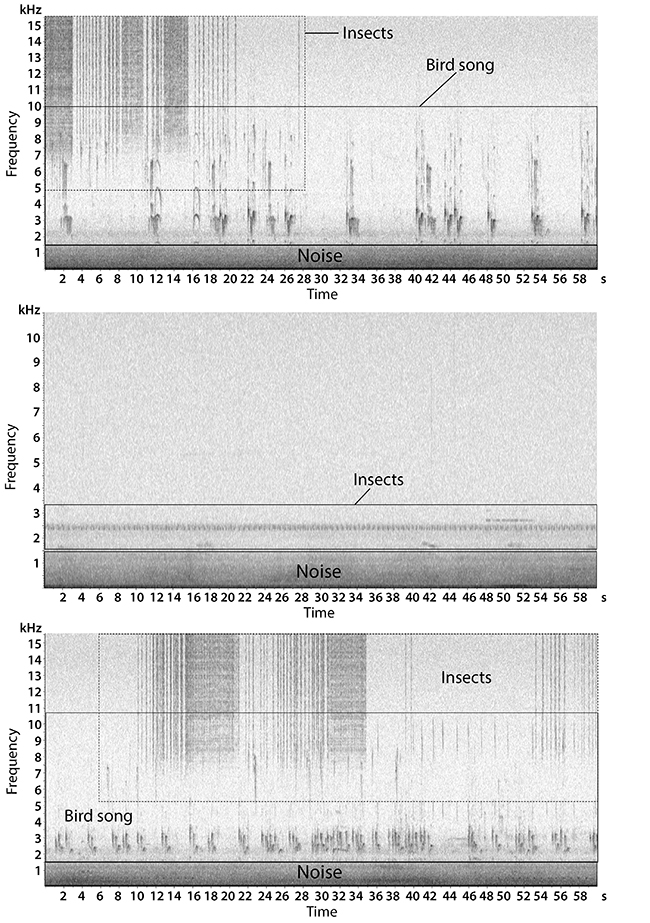
Figure 2. Spectrograms of the soundscape recorded at the border of the Pontifical Catholic University forest fragment, Belo Horizonte, Minas Gerais, Brazil.. Frequencies below 1550 Hz are filled by noise and biophony (insects, and bird songs) was concentrated between 1550 to 16000 Hz.
The soundscape of the study area was dominated by biophony and anthropogenic noise. Biophony was emitted by 12 potential insect species, 21 of bird taxa, one marmoset species (Callithrix penicillata) and three potential species of bats (Tables 1 and 2). The main noise sources affecting the area came from a small airport, vehicular traffic, and from the nearby university (Table 3). Some of the noises identified were: helicopters, airplanes, sirens, horns, motorcycles, sports games, lawnmowers and people’s conversations.
Biophony. ACI was significantly higher at the border than at the centre of the forest (F = 83.6, DF = 1, MS = 3.86, P < 0.01; Figure 3). No significant difference was found between the months sampled (F = 1708, DF = 2, MS = 0.145, P = 0.18).
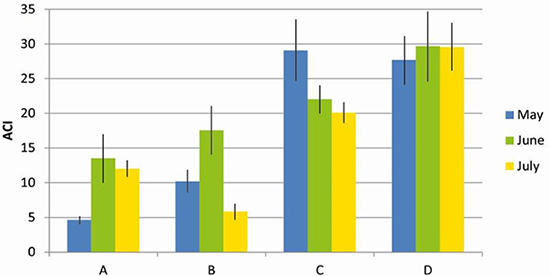
Figure 3. Biophony values (Acoustic Complexity Index - ACI) in the centre (A and B) and in the border (C and D) of the Pontifical Catholic University forest, Belo Horizonte, Minas Gerais, Brazil.
Biophony vs noise. The border of the forest fragment presented both higher noise values (i.e. PSD) and greater biophony (i.e. ACI) in relation to the centre. The ACI and PSD values presented a medium strength positive correlation, demonstrating that the higher the noise, the greater the biophony (r = 0.56; N = 96, P< 0.01; Figure 4).
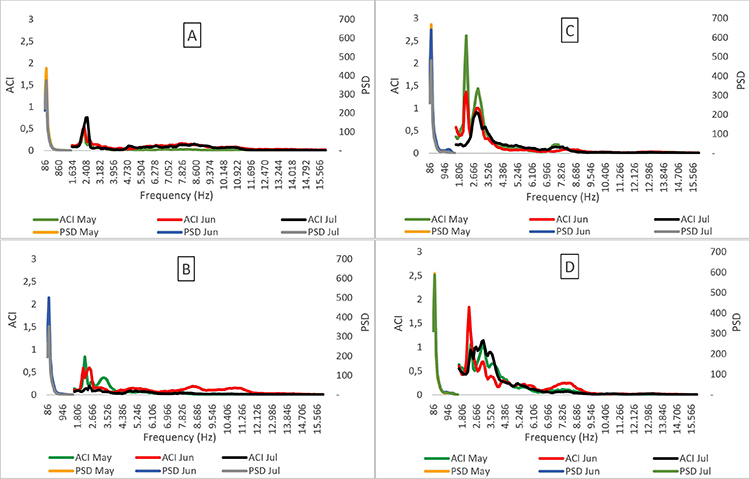
Figure 4 Biophony (ACI) and noise (PSD) values distributed across frequency bands for the sites in the centre (A and B) and border (C and D) of the Pontifical Catholic University forest, Belo Horizonte, Minas Gerais, Brazil.
Potential species richness. Acoustic diversity of sound types was found to be higher at sampling points in the centre of the forest fragment (Table 2).
Table 1. Potential number of species at border and centre sites of an urban tropical forest fragment in Belo Horizonte, Minas Gerais, Southeast Brazil.
Table 2. Bird species recorded by passive acoustic monitoring at an urban forest fragment in Minas Gerais, Southeast Brazil. NI, unidentifed species.
Noise – Equivalent levels (Leq). As expected, noise levels were higher on the border (Leq max 70 dB (Z)) than in the central area (Leq max 64.1 dB (Z); Table 3).
Table 3. Mean noise (sound pressure) level at the border and the centre of an urban tropical forest fragment in Belo Horizonte, Minas Gerais, Southeast Brazil.
Noise – Power Spectral Density (PSD). Accordingly, PSD values at the border were significantly higher than in the centre of the forest fragment (F = 85.67, DF = 1, MS = 0.657, P <0.01; Figure 5). PSD varied between months both in the centre and in the border, being significantly lower in July (F = 5.94, DF = 2, MS = 0.07, P <0.05).
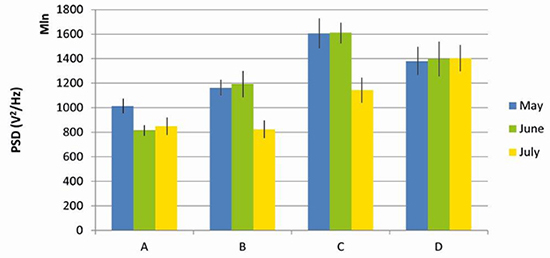
Figure 5. Noise values (Power spectral density - PSD) in the centre (A and B) and in the border (C and D) of the Pontifical Catholic University forest fragment, Belo Horizonte, Minas Gerais, Brazil.
Our findings can be interpreted as changes in the acoustic behaviour of species due to noise, but they could also be the result of species segregation by degree of vocal plasticity (i.e. ability to adapt) or even of differences in composition of communities.Our results showed that background noise is almost 6 dB (Leq) higher at the border points. In acoustics, an increase of 3 dB doubles the sound intensity (Rossing, 2007). Higher noise levels at the border of the forest were expected due to the vicinity of sound sources, such as streets and buildings. Other studies have obtained similar results in Southeast Brazil, recording higher noise values on the border of an urban park (Duarte et al., 2011) and in an Atlantic forest fragment (Duarte et al., 2015; 2017). This result may be due to many factors, for example, higher abundance of individuals at the border, or an attempt by animals to use compensatory mechanisms to communicate in noisy areas.
Since we found a higher number of sound types in the centre of the forest, which suggests higher species richness, it is likely that the animal community at the border has changed its vocal behaviour by increasing amplitude, and/or the calling rate and duration of their calls as a form of adaptation to compensate for the noise. Studies with birds and mammals have shown that animals are able to increase the amplitude of their vocalizations in noisy areas; this is known as the Lombard Effect (Cynx et al., 1998; Brumm et al., 2004; Brumm & Slater, 2006). Our previous study on the effects of mining truck traffic on cricket calling activity showed that species in noisy sites emit calls with higher average power, an indicator of sound intensity levels (Duarte et al., 2019).
Since the ACI uses the variation of amplitude in each frequency band to calculate biophony, the increase in the amplitude of vocalizations can generate higher values of this index (Zhao et al. 2019). Other types of noise compensatory mechanisms such as repetition of notes, syllables, increased call rate might also influence the values of the ACI (Brumm et al., 2004; Sun & Narins, 2005). Other studies have found animals exhibiting greater acoustic activity in noisier areas. Pieretti & Farina (2013) showed that both ACI values and noise were significantly higher with increasing proximity to a road, suggesting a more active singing/vocalising community in those sites where noise was more intense. Duarte et al., (2015) also found higher ACI values throughout the day in a noisy area, despite having less species compared to a silent area, indicating the possible use of compensatory mechanisms to communicate in presence of noise.
The direct energetic cost of vocal behaviour includes the energy required to produce the sound as well as the energy lost by not feeding during the time spent in vocalizations (Deecke et al., 2005). However, the indirect costs of acoustic communication include the possibility of passing information to unwanted receivers, such as competitors (Hammond & Bailey 2003), predators (Hosken et al., 1994; Mougeot & Bretagnolle, 2000) and parasites (Muller & Robert, 2002). A study found that an urban bird can vocalize up to 70 dB in response to varying noise levels (Díaz et al., 2011). However, vocal activity declined sharply above the threshold of 70 dB, which suggests that this strategy is costly for birds. This study further suggests that bird populations in noisy environments, such as cities, may face a greater challenge for survival compared to those in quiet areas, even for species that can mitigate the interference of urban noise in their acoustic communication (Díaz et al., 2011). Thus, in our study it is possible that animals were spending excessive energy in their communication.
Pieretti & Farina (2013) consider the possibility of birds remaining segregated between noisy and silent areas based on their vocal plasticity and on the ability to increase their song production. Thus, individuals genetically predisposed to produce intense vocalization behaviour can tolerate anthropogenic noise and live in noisy places, while others that are more sensitive to noise prefer to live in quieter areas. It might be possible that the animals that vocalized on the border of our study site were more noise tolerant and thus continued vocalizing, despite high levels of anthropogenic noise. However, it is also possible that the higher biophony at the border was related to a difference in animal community compositions between the border and the centre of the forest. Greater species richness singing at the border would also be responsible for a greater acoustic activity at the border. However, we found higher diversity of sound types in the centre of the forest, where biophony was lower. Similar results were found close to an open-cast mine in Brazil (Duarte et al., 2015). Studies on the edge effect in a community of birds indicate that there is a tendency for lower species richness and density at the edges of forests (Kroodsma, 1982; Laurance, 2004). Perillo et al., (2017) found lower bird species richness in areas with higher noise levels in urban green areas in Belo Horizonte, Minas Gerais, Brazil. These evidences do not support the hypothesis that higher levels of biophony in our border sites might be linked to higher species richness or abundance.
In our study, we registered lower noise levels in July compared to other months. Indeed, this month corresponds to the university’s major vacation period, with less students attending the nearby sport centre and, consequently, less noise produced by cars and buses, whistles, sport noises and people talking. Contrarily, biophony values did not vary over the three months of data collection. Since the data were collected during three months of the same season (dry), we did not expect variation in acoustic activity of the animals during the months studied.
Our findings showed that the impact of noise on the fauna can be acoustically monitored by measuring altered biophonic activity. Our results show how the relation between biophony, soniferous species richness, and anthropogenic noise is complex, mainly at a local scale. We suggest the use of ecoacoustics for future studies since it can be useful for interpreting anthropogenic disturbances and help the development of adequate urban management and conservation strategies.
We are grateful to Marina Scarpelli and Miguel Cançado for their help during data acquisition. We thank also the anonymous referees for their comments and suggestions on this manuscript. The authors would like to thank the Brazilian Council for Scientific and Technological Development (CNPq) for their continuing support. RJY was financially supported by CNPq and the Minas Gerais Research Foundation (FAPEMIG - PPM). S.G.S was supported by a FAPEMIG postgraduate scholarship and M.H.L.D was supported by the Brazilian Coordination for the Improvement of Higher Education Personnel (CAPES - PNPD20131384) during this research. This study was funded competitively by FAPEMIG from a financial donation made by VALE.
Barber, J. R., Crooks, K. R. & Fistrup, K. M. (2009). The costs of chronic noise exposure for terrestrial organisms. Trends in Ecology and Evolution, 25, 180-189.
https://doi.org/10.1016/j.tree.2009.08.002
Berger-Tal, O., Wong, B.B.M, Candolin, U. & Barber J. (2019). What evidence exists on the effects of anthropogenic noise on acoustic communication in animals? A systematic map protocol. Enviromental Evidence, 8, 18-25.
https://doi.org/10.1186/s13750-019-0165-3
Bonier, F., Martin, P.R. & Sheldon, K.S. (2007). Sex-specific consequences of life in the city. Behavioral Ecology, 18, 121-129.
https://doi.org/10.1093/beheco/arl050
Brumm, H., Voss, K., Kollmer, I. & Todt, D. (2004). Acoustic communication in noise: regulation of call characteristics in a New World monkey. Journal of Experimental Biology, 207, 443-448.
https://doi.org/10.1242/jeb.00768
Brumm, H. (2006). Signaling through acoustic windows: nightingales avoid interspecific competition by short-term adjustment of song timing. Journal of Comparative Physiology, 12, 1279-1285.
https://doi.org/10.1007/s00359-006-0158-x
Brumm, H. & Slater, P.J.B. (2006). Animals can vary signal amplitude with receiver distance: evidence from zebra finch song. Animal Behaviour, 71, 699-705.
https://doi.org/10.1016/j.anbehav.2006.01.020
Brumm, H. & Zollinger, S., A. (2011). The evolution of the Lombard effect: 100 years of psychoacoustic research. Behaviour, 148, 1173-1198
https://doi.org/10.1163/000579511X605759
Cynx, J., Lewis, R., Tavel, B. & Tse, H. (1998). Amplitude regulation of vocalizations in noise by a songbird Taeniopygia guttata. Animal Behaviour, 56, 107-113.
https://doi.org/10.1006/anbe.1998.0746
Deecke, V. B., Ford, J. K. B. & Slater, P. J. B. (2005). The vocal behaviour of mammal-eating killer whales: communicating with costly calls. Animal. Behaviour, 69, 395-405.
https://doi.org/10.1016/j.anbehav.2004.04.014
Díaz, M.; Parra, A. & Gallardo C. (2011). Serins respond to anthropogenic noise by increasing vocal activity. Behavioral Ecology, 22, 332-336.
https://doi.org/10.1093/beheco/arq210
Duarte, M. H. L., Vecci, M. A., Hirsch A. & Young R. J. (2011). Noisy human neighbours affect where urban monkeys live. Biology Letters, 7, 840-842.
https://doi.org/10.1098/rsbl.2011.0529
Duarte, M.H.L., Kaizer, M.C., Young, R.J., Rodrigues M. & Sousa-Lima, R.S. (2017). Mining noise affects loud call structures and emission patterns of wild black-fronted titi monkeys. Primates, 59, 89-97.
https://doi.org/10.1007/s10329-017-0629-4
Duarte, M.H.L., Sousa-Lima, R.S, Young, R.J. Farina, A., Vasconcelos, M., Rodrigues, M. & Pieretti, N. (2015). The impact of noise from open-cast mining on Atlantic forest biophony. Biological Conservation, 191, 623-631.
https://doi.org/10.1016/j.biocon.2015.08.006
Duarte, M.H.L., Caliari, E.P., Scarpelli, M.D., Lobregat, G.O., Young,R.J., & Sousa-Lima, R.S. (2019). Effects of mining truck traffic on cricket calling activity. The Journal of the Acoustical Society of America, 146, 656-664.
https://doi.org/10.1121/1.5119125
Farina, A., Pieretti, N. & Piccioli, L. (2011a). The soundscape methodology for long-term bird monitoring: A Mediterranean Europe case-study. Ecological Informatics, 6, 354-363.
https://doi.org/10.1016/j.ecoinf.2011.07.004
Farina, A., Lattanzi, E., Malavasi, R., Pieretti, N. & Piccioli, L. (2011b). Avian soundscapes and cognitive landscapes: theory, application and ecological perspectives. Landscape Ecology, 26, 1257-1267.
https://doi.org/10.1007/s10980-011-9617-z
Hammond, T. J. & Bailey, W. J. (2003). Eavesdropping and defensive auditory masking in an Australian bushcricket, Caedicia (Phaneropterinae: Tettigoniidae: Orthoptera). Animal Behaviour, 140, 79-95.
https://doi.org/10.1163/156853903763999917
Mougeot, F. & Bretagnolle, V. (2000). Predation as a cost of sexual communication in nocturnal seabirds: an experimental approach using acoustic signals. Animal Behaviour, 60, 647-656.
https://doi.org/10.1006/anbe.2000.1491
Muller, P., & Robert, D. (2002). Death comes suddenly to the unprepared: singing crickets, call fragmentation, and parasitoid flies. Behavioral Ecology, 13, 598-606.
https://doi.org/10.1093/beheco/13.5.598
Nemeth, E., and Brumm, H. (2009). Blackbirds sing higher-pitched songs in cities: adaptation to habitat acoustics or side-effect of urbanization? Animal Behaviour, 78, 637-641.
https://doi.org/10.1016/j.anbehav.2009.06.016
Perillo, A., Mazzoni, L.G., Passos,L.F., Goulart,V. D. L. R., Duca, C. & Young, R.J. (2017). Anthropogenic noise reduces bird species richness and diversity in urban parks, Ibis, 159, 638–646.
https://doi.org/10.1111/ibi.12481
Pieretti, N., Farina, A. & Morri, D. (2011). A new methodology to infer the singing activity of an avian community: The Acoustic Complexity Index (ACI). Ecological Indicators, 11, 868-873.
https://doi.org/10.1016/j.ecolind.2010.11.005
Pieretti, N. & Farina, A. (2013). Application of a recently introduced index for acoustic complexity to an avian soundscape with traffic noise. The Journal of the Acoustical Society of America, 134, 891.
https://doi.org/10.1121/1.4807812
Pieretti, N., Duarte, M.H.L., Sousa-Lima, R.S., Rodrigues, M., Young, R.J. & Farina, A. (2015). Determining temporal sampling schemes for passive acoustic studies in different tropical ecosystems.Tropical Conservation Science, 8, 215–234.
https://doi.org/10.1177/194008291500800117
Pijanowski, B. C., Villanueva-Rivera, L. J., Dumyahn, S. L., Farina, A., Krause, B. L. Napoletano, B. M., Gage, S. H. & Pieretti, N. (2011). Soundscape Ecology: The science of sound in the landscape. Bioscience, 61, 203-216.
https://doi.org/10.1525/bio.2011.61.3.6
Rossing, T.D. (2007). Springer Handbook of Acoustics. NewYork: Springer-Verlag New York Inc. 1182 pp.
https://doi.org/10.1007/978-0-387-30425-0
Santos, S.G., Duarte, M.H.L., Sousa-Lima, R.S. & Young, R.J. (2017). Comparing contact calling between black tufted-ear marmosets (Callithrix penicillata) in a noisy urban environment and in a quiet forest. International Journal of Primatology, 38, 1130-1137.
https://doi.org/10.1007/s10764-017-0002-x
Schafer, R. M. (1977). The Tuning of the World. Knopf. Michigan University.
Sjölander, K. & Beskow, J. (2000). Wavesurfer: an open source speech tool. Interspeech, 464-467.
Slabbekoorn, H. & Peet, M. (2003). Birds sing at a higher pitch in urban noise. Nature, 424, 267.
https://doi.org/10.1038/424267a
Slabbekoorn, H. & Ripmeester, E. A. (2008). Birdsong and anthropogenic noise: implications and applications for conservation. Molecular Ecology, 17, 72-83.
https://doi.org/10.1111/j.1365-294X.2007.03487.x
Sueur, J. & Farina, A. (2015). Ecoacoustics: the ecological investigation and interpretation of environmental sound. Biosemiotics, 8,493–502.
https://doi.org/10.1007/s12304-015-9248-x
Sun, J. W. C. & Narins, P. M. (2005). Anthropogenic sounds differentially affect amphibian call rate. Biological Conservation, 121, 419-427.
https://doi.org/10.1016/j.biocon.2004.05.017
Teixeira, B., Hirsch,A. Goulart,V. D. L. R., Passos,L., Teixeira,C.P. James, P. & Young, R.J.(2015). Good neighbours: distribution of black-tufted marmoset (Callithrix penicillata) in an urban environment. Wildlife Research, 42, 579-589.
https://doi.org/10.1071/WR14148
Tolentino, V. C. D. M, Baesse, C. Q. & Melo, C. D. (2018). Dominant frequency of songs in tropical bird species is higher in sites with high noise pollution. Environmental Pollution, 235, 983-992.
https://doi.org/10.1016/j.envpol.2018.01.045
Vasconcelos, M. F., Mazzoni, L. G., Perillo, A.; Guerra, T., Morais, R., Garzon, B., Santos, J. E., Guimarães, L. S. L., Oliveira, Almeida, T., Peixoto, H. J. C., Dutra, E. C., Pedroso, L. F., Valério, F. A., Petrocchi, D., Santos, L. P. S. Dias, J. E. M., Morais, S. A., Garcia, F. I. A., Benfica, C. E. R. T. & Ribeiro, B. P. (2013). Long-term avifaunal survey in an urban ecosystem from southeastern Brazil, with comments on range extensions, new and disappearing species. Papéis Avulsos de Zoologia da Universidade de São Paulo, 53, 327-344.
https://doi.org/10.1590/S0031-10492013002500001
Warren, P. S., Katti, M., Ermann, M. & Brazel, A. J.. (2006). Urban bioacoustics: it’s not just noise. Animal Behaviour, 71, 491-502.
https://doi.org/10.1016/j.anbehav.2005.07.014
Zhao, Z., Xu, Z. Yong, Bellisario, K., Zeng, R. Wen, Li, N., Zhou, W. Yang, & Pijanowski, B. C. (2019). How well do acoustic indices measure biodiversity? Computational experiments to determine effect of sound unit shape, vocalization intensity, and frequency of vocalization occurrence on performance of acoustic indices. Ecological Indicators, 107.
https://doi.org/10.1016/j.ecolind.2019.105588
* Autor para correspondencia
1Pontifical Catholic University of Minas Gerais, Belo Horizonte, Minas Gerais, Brazil
2Polytechnic University of Marche, Ancona, Italy
3University of Salford, Manchester, United Kingdom