Recibido: 8 de mayo de 2020; Aceptado: 9 de noviembre de 2020
Abstract
We describe the advertisement call of seven males and observations on the natural history related to the parental attention by males, of a population of nurse frogs attributed to the Leucostethus fraterdanieli complex from the Central Cordillera in the Department of Caldas, Colombia. The advertisement call consists of a long sequence of repeated (x̅ = 143 notes.min-1) pulsed notes, which have an average duration of 79 ± 17 ms (range 63-122 ms), separated by time intervals between 187-413 ms (289 ± 67 ms). The dominant frequency values are in the range between 3209-3520 Hz. Our results suggest differences in temporal and spectral parameters between the population studied and other populations assigned to L. fraterdanieli, L. brachistriatus, and the recently described L. jota.
Key words:
Amphibia, Andes, Bioacoustics, Caldas, Colombian Central Cordillera, Male brooding behavior, Phytotelmata..Resumen
Describimos el canto de advertencia de una población atribuida a Leucostethus fraternadieli de los Andes centrales de Colombia, basados en el análisis temporal y espectral del canto de siete machos, y aportamos observaciones de historia natural asociadas al cuidado parental de los machos. El canto de advertencia consiste en una larga secuencia de notas pulsadas repetidas (x̅ = 143 notes.min-1), las cuales tienen una duración promedio de 79 ∓ 17 ms (rango 63-122 ms), separadas por intervalos de tiempo de entre 187-413 ms (289 ∓ 67 ms). Los valores de frecuencia dominante registrados están en el rango entre 3209-3520 Hz. Nuestros resultados sugieren diferencias en parámetros temporales y espectrales entre la población estudiada y otras poblaciones asignadas a L. fraterdanieli, L. brachistriatus, y la especie recientemente descrita L. jota.
Palabras clave:
Amphibia, Andes, Atención de nidadas, Bioacústica, Caldas, Cordillera Central de Colombia, Fitotelmata..Introduction
Bioacoustics provide an useful instrument in taxonomy and systematics for Neotropical anuran species (Toledo et al., 2015; Köhler et al., 2017), particularly for poison frogs (Dendrobatoidea) (Lötters et al., 1999; 2007; Brown et al. 2008a; Twomey & Brown, 2008; Brown et al., 2011), with newly described taxa usually including detailed call descriptions (e.g. Myers et al., 1998; Brown et al., 2008a; Márquez et al., 2017). For closely related but taxonomically uncertain species, bioacoustic information is also relevant, since call parameters can show differences that morphology does not (Angulo & Reichle, 2008; Castroviejo-Fisher et al., 2008; Padial et al., 2008; Pritti et al., 2016). Also, natural history traits related to microhabitat selection for breeding and parental care have shown interesting differences in systematics and evolutionary terms between closed aposematic resembling dendrobatids (e.g. Brown et al., 2008 b, c). However, knowledge about the ecology and natural history for non-aposematic dendrobatid frogs is still scarce, and the lack of such information has limited systematic proposals for the evaluation of morphological, genetic and bioacoustic characters.
Within the Dendrobatidae, the genus Leucostethus comprises at least six species (L. argyrogaster, L. brachistriatus, L. fraterdanieli, L. fugax, L. jota and L. ramirezi) of South American endemic nurse frogs, with the highest diversity in the Northern Andes of Colombia (Grant et al., 2017; Frost, 2020), and with suspectedcryptic diversity. For example, Grant et al. (2017) found five clades that seem to represent different species within the fraterdanieli complex. Among them, there was a clade distributed along the Cauca river valley and the adjacent flanks of the Central and Western Cordilleras of Colombia, from Popayán (Department of Cauca) to Filandia (Department of Quindío), that they assigned to C. brachistriatus (Rivero & Serna, 1986). As pointed out by Grant & Castro (1998), the extensive within- and among-population morphological variation in L. fraterdanieli (previously included in the genus Colostethus and transferred to Leucostethus by Marín et al., 2018), suggests a complex of closely resembling species, corroborated by different authors (Grant et al., 2006, 2017; Santos et al., 2009; Marín et al., 2018). Therefore, hereafter we will refer to our study species as L. fraterdanieli complex.
The Leucostethus fraterdanieli complex (Silverstone, 1971) comprises a group of small nurse frogs (Dendrobatidae: Colosthetinae) endemic of the sub-Andean and Andean forests in the Central and Western cordilleras of Colombia, in an elevational range between 1100 and 2500 m a.s.l. (Silverstone, 1971; Rivero & Serna, 1995; Grant & Castro, 1998; Acosta-Galvis, 2000; Bernal et al., 2005; Bernal & Lynch, 2008; Castro-Herrera & Vargas-Salinas, 2008; Sánchez, 2013; Rojas-Morales et al., 2014; Guevara-Molina et al., 2017). Bernal & Lynch (2008) indicated a record at 650 m of elevation, but that appears unlikely to us, as deducted from their data table (see Appendix 1 in Bernal & Lynch, 2008).
The Leucostethus fraterdanieli complex includes diurnal-terrestrial frogs that are active near creeks and streams inside primary and secondary forests, even in forest fragments of less than 1 ha, surrounded by urban and rural areas (Guevara-Molina et al., 2017). Although some behavioral aspects such as anurophagy (e.g. consumption of Pristimantis achatinus) have been reported (Cárdenas-Ortega & Herrera-Lopera, 2016), other traits of their natural history, such as microhabitat use and reproductive behavior, are still unknown. In this study, we describe the advertisement call of a population assigned to the L. fraterdanieli complex, based on individuals from an urban locality in the Department of Caldas, Central Cordillera of Colombia. In addition, we provide information on natural history, such as calling and oviposition site, and egg-attendance and embryo transport behavior displayed by males.
Materials and methods
Study site. Call recordings were made at the Botanical Garden of the University of Caldas, Municipality of Manizales, Department of Caldas, Colombia (05°3’26.68”N, 75°29’45.28”W, WGS84, 1960 m a.s.l), which is a forested area in the central-southern part of Manizales, containing a representation of different types of vegetation, such as native secondary forest, coniferous forest (mainly consisting of Pinus patula), and crop areas for agricultural experimentation. The vegetation and environmental conditions in this area correspond to the moist lower montane forest (or bmh-MB) in Holdridge (1982) life zone system, with an annual rainfall average of 2600 mm, increasing in March-May and September-November (Corpocaldas, 2012). Annually, the warmest months are May and July with average temperatures of 24 °C, while the coldest month is September with an average of 11.4 °C (Corpocaldas, 2012). Native flora from the forests where L. fraterdanieli complex has been recorded, include cedro negro (Juglans neotropica, Juglandaceae), encenillo (Weinmannia pubescens, W. elliptica, Cunoniaceae), yarumo blanco (Cecropia telealba, Urticaceae), dulomoco (Saurauia cuatrecasana, Actinidaceae), drago (Croton magdalenensis, Euphorbiaceae), nigüito (Miconia poecilantha, M. theaezans, Melastomataceae), wax palm (Ceroxylon quindiuense, Arecaceae), among many others (Vargas, 2002).
Call analysis. The advertisement call of seven individuals of the L. fraterdanieli complex is described based on recordings made by the first author on 12 May 2012 between 14:33-17:32 h. For the description, 84 calls in total (12 calls for each male), randomly selected from one minute of recording for each individual were analyzed. Males were recorded at 50-70 cm average distance with a Sennheiser© ME64 cardioid unidirectional microphone, adapted to a Marantz© PMD620 digital recorder. Temperature (Table 1) was registered at the time of each recording, with an Extech© EA25 infrared thermometer, and male’s snout-to-vent length (SVL) was measured with a 0.01 mm Mitutoyo© MI-500-171-30B precision’s caliper (Table 1). Because the individuals of L. fraterdanieli complex have shown an aggregate local distribution, the recordings were taken at least 5-20 m apart for each male, to avoid pseudoreplication. No voucher specimens were collected in this study; however, 13 vouchers from this locality are housed at the herpetological collection of the Museo de Historia Natural, Universidad de Caldas (MHN-UCa 416-418, 447-452, 831-834). We followed Wells (2007), Toledo et al. (2015) and Köhler et al. (2017) for the description of the temporal and spectral parameters of the calls. Recordings were digitized and analyzed with the software RAVEN © Pro 1.4 (Bioacoustics Research Program 2011) at a resolution of 16 bits and 44.1 kHz sampling rate. Oscillograms and spectrograms were analyzed with a FFT (Fast Fourier Transformation) of 512 points and Hann algorithm. Description of the call parameters are presented as mean ∓ standard deviation (range). During the course of other research (2010-2012), the first author gathered additional information on microhabitat and specific observations on the behavior and activity of wild individuals in areas close to that of this study (municipalities of Neira and Villamaría, Department of Caldas).
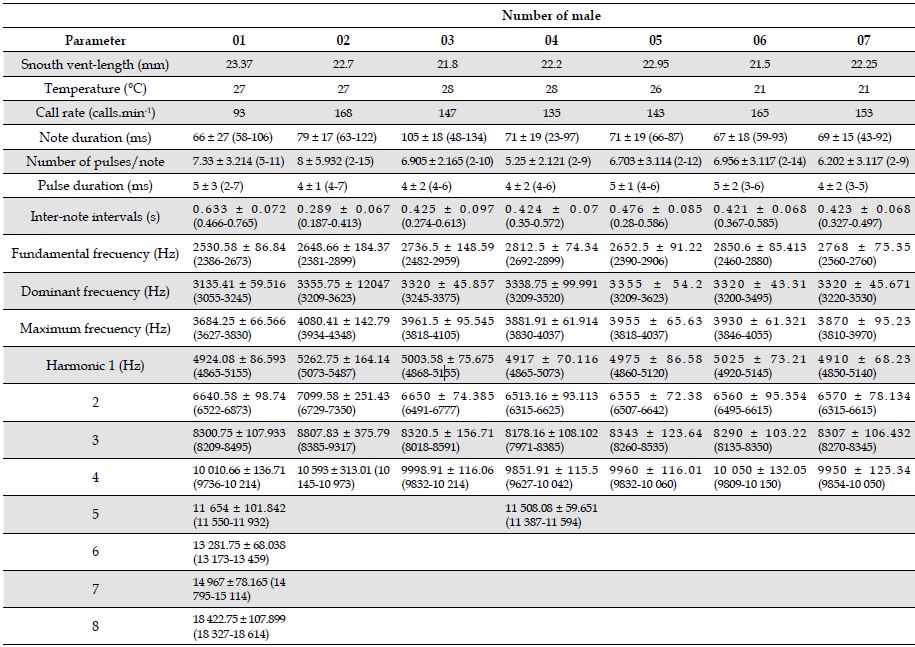
Table 1: Temporal and spectral traits of the advertisement call of seven males of the Leucostethus fraterdanieli complex from Manizales, Caldas, Colombia. Snouth vent-length (SVL) of males and environmental temperature (°C) are given. Values correspond to the mean ± standard deviation (range).
Number of male
Parameter
01
02
03
04
05
06
07
Snouth vent-length (mm)
23.37
22.7
21.8
22.2
22.95
21.5
22.25
Temperature (°C)
27
27
28
28
26
21
21
Call rate (calls.min-1)
93
168
147
135
143
165
153
Note duration (ms)
66 ± 27 (58-106)
79 ± 17 (63-122)
105 ± 18 (48-134)
71 ± 19 (23-97)
71 ± 19 (66-87)
67 ± 18 (59-93)
69 ± 15 (43-92)
Number of pulses/note
7.33 ± 3.214 (5-11)
8 ± 5.932 (2-15)
6.905 ± 2.165 (2-10)
5.25 ± 2.121 (2-9)
6.703 ± 3.114 (2-12)
6.956 ± 3.117 (2-14)
6.202 ± 3.117 (2-9)
Pulse duration (ms)
5 ± 3 (2-7)
4 ± 1 (4-7)
4 ± 2 (4-6)
4 ± 2 (4-6)
5 ± 1 (4-6)
5 ± 2 (3-6)
4 ± 2 (3-5)
Inter-note intervals (s)
0.633 ± 0.072 (0.466-0.765)
0.289 ± 0.067 (0.187-0.413)
0.425 ± 0.097 (0.274-0.613)
0.424 ± 0.07 (0.35-0.572)
0.476 ± 0.085 (0.28-0.586)
0.421 ± 0.068 (0.367-0.585)
0.423 ± 0.068 (0.327-0.497)
Fundamental frecuency (Hz)
2530.58 ± 86.84 (2386-2673)
2648.66 ± 184.37 (2381-2899)
2736.5 ± 148.59 (2482-2959)
2812.5 ± 74.34 (2692-2899)
2652.5 ± 91.22 (2390-2906)
2850.6 ± 85.413 (2460-2880)
2768 ± 75.35 (2560-2760)
Dominant frecuency (Hz)
3135.41 ± 59.516 (3055-3245)
3355.75 ± 12047 (3209-3623)
3320 ± 45.857 (3245-3375)
3338.75 ± 99.991 (3209-3520)
3355 ± 54.2 (3209-3623)
3320 ± 43.31 (3200-3495)
3320 ± 45.671 (3220-3530)
Maximum frecuency (Hz)
3684.25 ± 66.566 (3627-3830)
4080.41 ± 142.79 (3934-4348)
3961.5 ± 95.545 (3818-4105)
3881.91 ± 61.914 (3830-4037)
3955 ± 65.63 (3818-4037)
3930 ± 61.321 (3846-4055)
3870 ± 95.23 (3810-3970)
Harmonic 1 (Hz)
4924.08 ± 86.593 (4865-5155)
5262.75 ± 164.14 (5073-5487)
5003.58 ± 75.675 (4868-5155)
4917 ± 70.116 (4865-5073)
4975 ± 86.58 (4860-5120)
5025 ± 73.21 (4920-5145)
4910 ± 68.23 (4850-5140)
2
6640.58 ± 98.74 (6522-6873)
7099.58 ± 251.43 (6729-7350)
6650 ± 74.385 (6491-6777)
6513.16 ± 93.113 (6315-6625)
6555 ± 72.38 (6507-6642)
6560 ± 95.354 (6495-6615)
6570 ± 78.134 (6315-6615)
3
8300.75 ± 107.933 (8209-8495)
8807.83 ± 375.79 (8385-9317)
8320.5 ± 156.71 (8018-8591)
8178.16 ± 108.102 (7971-8385)
8343 ± 123.64 (8260-8535)
8290 ± 103.22 (8135-8350)
8307 ± 106.432 (8270-8345)
4
10 010.66 ± 136.71 (9736-10 214)
10 593 ± 313.01 (10 145-10 973)
9998.91 ± 116.06 (9832-10 214)
9851.91 ± 115.5 (9627-10 042)
9960 ± 116.01 (9832-10 060)
10 050 ± 132.05 (9809-10 150)
9950 ± 125.34 (9854-10 050)
5
11 654 ± 101.842 (11 550-11 932)
11 508.08 ± 59.651 (11 387-11 594)
6
13 281.75 ± 68.038 (13 173-13 459)
7
14 967 ± 78.165 (14 795-15 114)
8
18 422.75 ± 107.899 (18 327-18 614)
Results
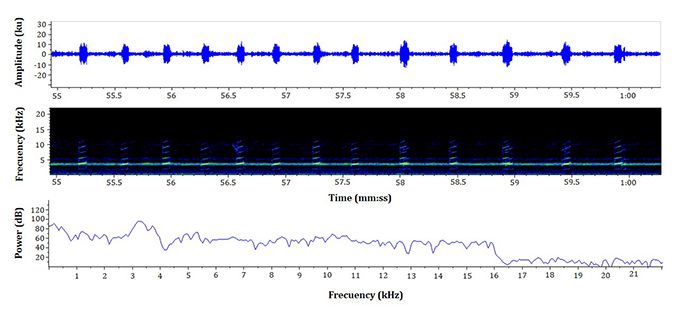
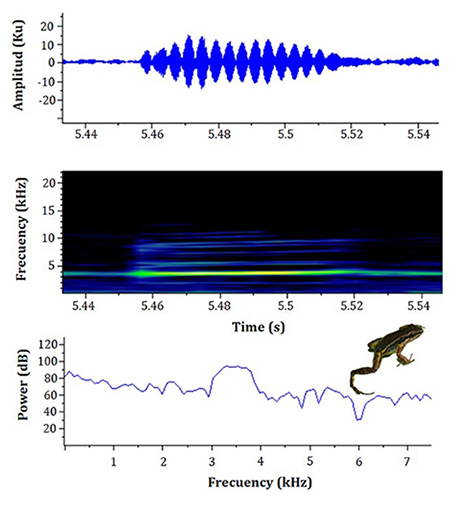
Advertisement call. Males of the L. fraterdanieli complex recorded in the study area called synchronically, overlapping during this activity. When a male began to emit advertisement calls, almost immediately neighbouring individuals also started to vocalize, forming a chorus audible at great distance (> 30 m). The advertisement call of L. fraterdanieli complex consisted of one note emitted in fast succession, with a mean call rate of 143 notes.min-1 (range 93-168; Table 1; Figure 1), each sounding like a ‘peep’ to the human ear. The notes had an average duration of 79 ∓ 17 ms (63-122 ms) and were separated by inter-note intervals of 289 ∓ 67 ms (187-413 ms). The dominant frequency of the call was 3338.75 ∓ 99.99 Hz (3209-3520 Hz); the minimum frequency 2812.5 ∓ 74.34 Hz (2692-2899 Hz) and the maximum frequency 3881.91 ∓ 69.91 Hz (3830-4037 Hz). All recorded males presented harmonic bands in their calls, but there were differences in the number of bands registered for each male (see Table 1). Harmonic 1 ranged from 4850 to 5487 Hz, harmonic 2 from 6315 to 7350 Hz, harmonic 3 from 7971 to 9317 Hz, harmonic 4 from 9736 to 10 973 Hz, harmonic 5 from 11 387 to 11 932 Hz. The harmonic 6 (13 173-13 459 Hz), harmonic 7 (14 795-15 114 Hz), and harmonic 8 (18 327-18 614 Hz) were present only in male 01. Notes were typically pulsed (2-11 pulses.note; Figure 2), although a small fraction of the notes did not have pulses. Pulses have a duration of 4 ∓ 0.5 ms (2-7 ms). Table 1 summarizes the acoustic attributes of the analyzed individuals.
Figure 1: Oscillogram, spectrogram, and power spectrum of the advertisement call of Leucostethus fraterdanieli (male 02 in Table 1, SVL 22.7 mm), from Manizales, Caldas, Colombia. Visualization of five seconds of recording; temperature 27 °C.
Figure 2: Oscillogram, spectrogram, and power spectrum of a pulsed note of an adult individual (male 06 in Table 1, SVL 21.5 mm) of the Leucostethus fraterdanieli complex from Manizales, Caldas, Colombia. Temperature, 21 °C.
Natural history observations. At the study site (Figure 3A), Leucostethus fraterdanieli complex is the most abundant diurnal amphibian species in the local assemblage. In a small area of 25 m2 in which two of the sound tracks were recorded, twelve individuals were counted calling hidden under the leaf litter of white yarumo trees (Cecropia telealba) and bamboos (Guadua angustifolia) (Figure 3B). In this site, on February 9, 2012, a male attending a clutch was found placed on a dry bromeliad leaf (Aechmea sp.) at 1.25 m above the ground, and approximately 40 m away from the nearest water body (Figure 4A). The male (SVL 23.7 mm) was in close contact with the egg mass performing a ‘brooding behavior’ sensuVockenhuber et al. (2008) (Figure 4B). The sex of the individual was determined based in the black spots in the throat and the swollen third finger, which are characteristics in adult males (Grant & Castro 1998). The egg-clutch contained 17 eggs in developmental stage 17 sensuGosner (1960) (Figure 4C). Both the male and the egg-clutch were monitored between 07:00-18:00 h, time during which the male remained attending eggs while staying on top of them. For other forested sites sampled near the cities of Manizales, Villamaría, and Neira, Department of Caldas, we observed that adults of the L. fraterdanieli complex were usually located at a distance (> 50 m) from water bodies during the day, with a calling activity from early morning (06:30-07:00 h) to the late afternoon (18:00-18:30 h). In contrast, we observed at night that parental males frequented leaf litter or rocks at the edge of small streams, carrying up to 15 larvae on their backs (N = 4, range 12-15 larvae; Figure 3C). Tadpoles aggregated at the edges of small streams and creeks (0.4-3.5 m width), with abundant sand and rocky soil (Figure 3D).
Figure 3: A, habitat of Leucostethus fraterdanieli complex at the Botanical Garden of the Universidad de Caldas, Manizales, Colombia, 2150 m a.s.l.; B, calling male concealed in a caulinar fallen leaf of bamboo (Guadua angustifolia) at this site; C, male carrying tadpoles on dorsum, before depositing them in a body of water, from the Chisperos creek, located near Manizales; D, tadpole at a margin of the Chisperos creek. Photographs by J. A. Rojas-Morales.
Figure 4: Oviposition site of individual of the Leucostethus fraterdanieli complex in the Botanical Garden of the Universidad de Caldas, Manizales, Colombia, 2150 m a.s.l. A, terrestrial bromeliad (Aechmea sp.) used as oviposition site; B, detail of the leaf where a male (SVL 23.7 mm) is attending a clutch in ventral brooding-behavior; C, egg-clutch with 17 embryos. Photographs by J. A. Rojas-Morales.
Discussion
Since anuran acoustic signals are species-specific and play a critical role in species recognition and mate choice (Wells, 2007), advertisement call analysis is a powerful tool to recognize distinct species. The advertisement call of the population of the L. fraterdanieli complex described here can be characterized as a long train of repeated ‘peep’ notes, with a maximum frequency up to 4000 Hz. In general terms, this call seems to be similar to that ‘chirp call’ type defined by Lötters et al. (2003). The call here described is similar in general patterns to that described by Grant & Castro (1998) as the advertisement call of L. brachistriatus (as C. fraterdanieli), based on two males collected within a cloud forest in the western slopes of the Cordillera Occidental, Department of Valle del Cauca. The call was described like a single note repeated up to 300 times, with frequencies between 3156-3250 Hz and harmonics occurring at 4800-6400 Hz (Individual 1), and 3593-3656 Hz and harmonics in the bands of 5500-7400 Hz (Individual 2; Grant & Castro, 1998). However, there is a difference in the note duration. For the population of the Western Cordillera, notes ranged between 38-49 ms (Grant & Castro, 1998), while for the population that we analyzed in the Central Andes, note duration was between 63-122 ms.
Recently, Marín et al. (2018) presented a bioacoustic comparison between L. fraterdanieli and L. jota from the Central Cordillera, in Antioquia. The call described for L. fraterdanieli includes values between 3100-3500 Hz, a rate of 98 ∓ 12.5 calls.min-1, with a call duration of 0.09 ± 0.03 s and intervals between calls 0.55 ± 0.16 s. These results are quite different in temporal traits from those of L. fraterdanieli complex from Manizales (call rate 98 vs 143 calls.min-1). Compared to L. jota, the advertisement call of the population of L. fraterdanieli complex presented here shows major differences in temporal parameters. A higher call duration (0.14 vs. 0.079 s), lower call rate (21.8 vs. 143 calls.min-1), and higher call interval (2.55 vs. 0.289 s) differentiates L. jota from L. fraterdanieli complex from Manizales. Guevara-Molina et al. (2017) presented information on the biology of L. fraterdanieli (L. brachistriatus sensuGrant et al., 2017), including an oscillogram and spectrogram of a call from the Department of Quindío, in the central Andes, but no additional details were provided.
Our results show that the advertisement call of the population of L. fraterdanieli complex from Manizales is quite different from other known Leucostethus (formerly within Colostethus) from the Upper Cauca river valley and the Central Cordillera (Grant & Castro, 1998; Marín et al., 2018). However, individuals from Manizales are morphologically indistinguishable from L. fraterdanieli sensu stricto (fide Grant et al., 2017). An integrative approach, including a molecular analysis and a study of larval morphology, is badly needed to assess the taxonomic status of this and other populations under the name of L. fraterdanieli in the Colombian Andes.
Life history data for Colostethinae frogs are limited (Wells, 1980; Coloma, 1995; Duellman 2004). Oviposition sites recorded in these frogs include shadow locations in mosses and under stones (Coloma, 1995). Parental care of the embryos and eggs are carried out by males; however, in species such as Colostethus panamensis, C. pratti and C. argyrogaster, the female is in charge of this function (Wells, 1980; Duellman, 2004). Despite this, microhabitat for oviposition is unknown for a large proportion of Colostethus and Leucostethus species. For L. fraterdanieli complex from Manizales, we observed one oviposition site located on a dead leaf of a terrestrial bromeliad (Figure 4A).
To our knowledge, this is the first reference for an oviposition site in Leucostethus. We also observed males performing parental care -i. e., egg-brooding- of an egg-clutch, and transporting larvae to the water, which show the role of males in the parental care of embryos until they continue their aquatic phase. However, reproductive activity and behavior patterns may vary from one population to another and from year to year (Wells, 1980); for that reason, is important to monitor this population over several years, in order to obtain data on territorial behavior, courtship, amplexus, and parental care of the eggs and embryos. The distinctive call, its preference for forested habitats and its abundance predicted through the calls, make this species a good model for bioacoustic monitoring to detect population trends in these Andean highly transformed ecosystems.